Problem Set 1: Buffers and Amino Acids
Opened: Saturday, 11 February 2023, 12:00 AM
Due: Saturday, 18 February 2023, 7:00 PM
View
Make a submission
General Instructions
- This is a group activity.
- Answer the problems shown below use the previously given pKa table as reference.
- Submit your answers in pdf/jpg formats only following naming, Chem40_PS1_<Section>_<Group No.>
- One submission per group is enough.
Problems
- What is the pH of a buffer prepared by adding 6.4g of NaOAc and 2.5ml HOAc to make 100ml of a buffer? The pKa of HOAc is 4.7. MW of NaOAC is 82 g/mol, while the MW of HOAc is 60 g/mol. Density of HOAc is 1.049. (OAc=acetate, HOAc=acetic acid)
- What is the pH of 0.1 M HCl? What is the pH of the same concentration of HOAc, Ka =
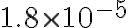 ?
? -
If you have 100 ml of a 0.10 M Tris buffer at pH 8.3 and you add 3.0 ml of 1M HCl, what will be the new pH of the solution? (Tris pKa= 8.3)
-
Name an amino acid in which the R group contains the following:–A hydroxyl group–An amide group–An acid group–A branched side chain–An aromatic ring
- Identify the nonpolar amino acids and the acidic amino acids in the following peptide: Glu-Thr-Val-Asp-Ile-Ser-Ala
-
Draw the structures of the following amino acids, indicating the charged form that exists at pH 4 and 10, respectively: H, N, W, P, Y
-
Sketch the titration curve for the amino acid cysteine and indicate the pKa values for all titratable groups. Also indicate the pH at which the zwitterion form predominates.